AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Lithium emission spectrum1/26/2024 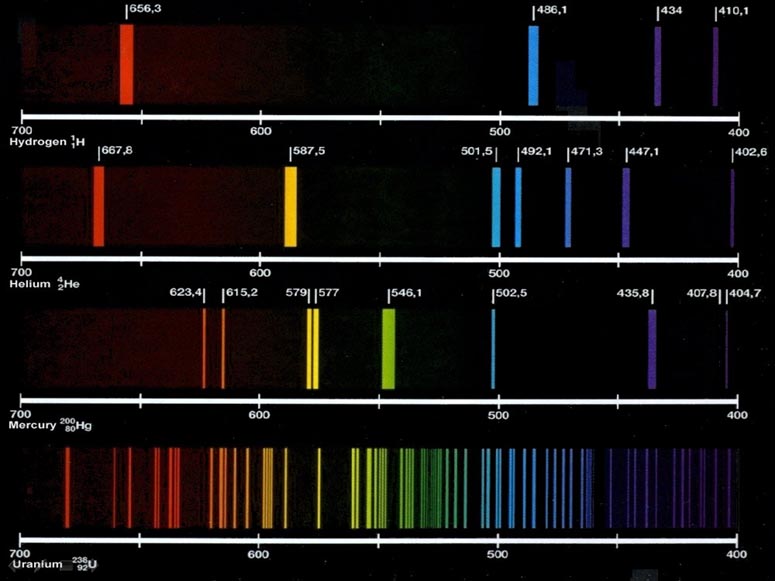
The idea of the test is that sample atoms evaporate, and since they are hot, they emit light when in flame. The test involves introducing a sample of the element or compound to a hot, non-luminous flame and observing the color of the flame that results. Process A flame test showing the presence of lithium. If the energy of the emitted photons is higher or lower than 380–700 nm, light will be emitted in the infrared, ultraviolet, or other regions of the electromagnetic spectrum that cannot be observed with the naked eye. If the photons are in the visible region of the light spectrum (wavelength 380–700 nm), a colored light emitted by the material is observed with the naked eye. In general, a larger transition between energy levels requires higher energy and will emit higher energy photons. This allows for the detection of different atoms. Therefore, as the electrons of different atoms fall to their unexcited energy levels, they will emit different amounts of energy, corresponding to lights of different wavelengths and frequencies. Electron excitation occurs at specific energy levels, indicating that the energy is quantized and corresponds to specific wavelengths (or frequencies). ĭifferent atoms or ions require different amounts of energy to promote one electron to one orbital higher than its normal state. As they fall back, they release energy in the form of photons, which are responsible for the light. When an atom or an ion absorbs the energy, electrons will jump from a lower energy level (the highest occupied molecular orbital, or HOMO at an unexcited state) to a higher energy level (the lowest occupied energy orbital, or LUMO at an excited state), and later fall back to their normal energy level (unexcited state). In flame tests, energy is emitted by the flame in the form of heat. Today, this low-cost method is used in secondary education to teach students to detect metals in samples qualitatively. In 1860, the unexpected appearance of sky-blue and dark red was observed in spectral emissions by Robert Bunsen and Gustav Kirchhoff, leading to the discovery of two alkali metals, caesium ( sky-blue) and rubidium (dark red). The Bunsen burner, combined with a prism (filtering the color interference of contaminants), led to the creation of the spectroscope, capable of emitting the spectral emission of various elements. Robert Bunsen invented the now-famous Bunsen burner in 1855, which was useful in flame tests due to its non-luminous flame that did not disrupt the colors emitted by the test materials. The characteristic bluish-green color of the flame is due to the copper. History The flame test carried out on a copper halide. The test provides qualitative data therefore, obtaining quantitative data requires subsequent techniques like flame photometry or flame emission spectroscopy. Safety precautions are crucial due to the flammability and toxicity of some substances involved. Wooden splints, Nichrome wires, cotton swabs, and melamine foam are suggested for support. The procedure uses different solvents and flames to view the test flame through a cobalt blue glass to filter the interfering light of contaminants such as sodium. 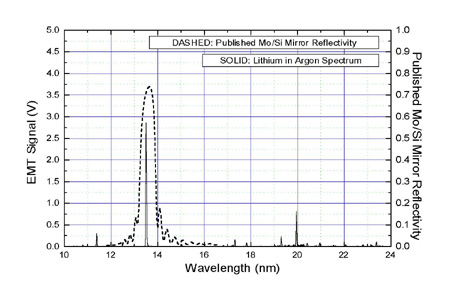 .jpg)
The color of the flames also generally depends on temperature and oxygen fed see flame colors. The color of the flames is understood through the principles of atomic electron transition and photoemission, where varying elements require distinct energy levels ( photons) for electron transitions. Process in chemistry to detect certain elementsĪ flame test, invented by Robert Bunsen, is a qualitative analysis technique used in chemistry to detect the presence of certain elements, primarily metal ions, based on each element's characteristic flame emission spectrum (which may be affected by the presence of chloride ions).
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
